|
Can you explain this answer? tests, examples and also practice JEE tests. Here you can find the meaning of The correct order of dipole moment of HF, H2S and H2Omolecules is in order.a)HF H2Oc)HF > H2S > H2Od)HF > H2O H2Oc)HF > H2S > H2Od)HF > H2O H2Oc)HF > H2S > H2Od)HF > H2O H2Oc)HF > H2S > H2Od)HF > H2O H2Oc)HF > H2S > H2Od)HF > H2O< H2SCorrect answer is option 'C'. H2S has the lowest dipole moment due to the less polar S-H bond. H2O has a slightly lower dipole moment than HF due to the less polar O-H bond. Dipole and quadrupole moment functions of the hydrogen halides HF, HCl, HBr, and HI: A Hirshfeld interpretation. This is because HF has the highest dipole moment due to the highly polar H-F bond. The correct order of dipole moment of HF, H2S, and H2O molecules is: HF > H2O > H2S. The dipole moment of H2O is 1.85 D, which is between that of HF and H2S. The O-H bond is highly polar because oxygen is highly electronegative. The dipole moment of H2S is 0.97 D, which is less than that of HF. The S-H bond is polar because sulfur is less electronegative than hydrogen. This makes sense because approximations were used in the calculations. As you can see the calculated dipole moments are much higher than the experimental. The total molecular dipole moment may be approximated as the vector sum of the individual bond dipole moments. page needed verification needed For polyatomic molecules, there is more than one bond. 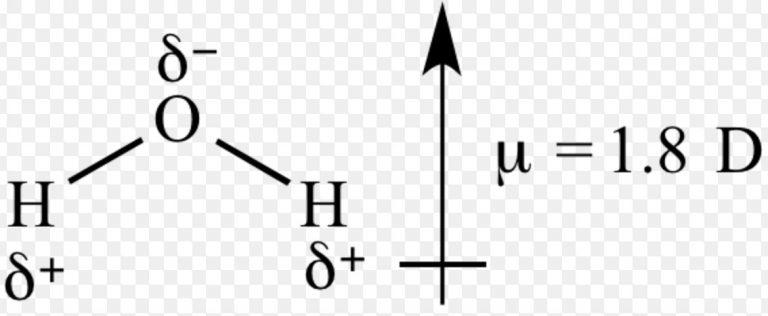 
Hydrogen sulfide (H2S) is also a polar molecule. The table below shows the calculated and experimental dipole moments of HF. 2, has zero dipole moment, while near the other extreme, gas phase potassium bromide, KBr, which is highly ionic, has a dipole moment of 10.41 D. The dipole moment of HF is 1.91 D, where D is the unit of dipole moment. B, linear model with consecutively antiparallel HF mole- cules dipole moment either zero or 1.91 C, linear model deduced from electron diffraction data. The H-F bond is highly polar because fluorine is highly electronegative. Hydrogen fluoride (HF) is a polar molecule. The dipole moment of a molecule is defined as the product of the magnitude of the charge on either end of the bond and the distance between the charges.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
